|
|
|
|
|
|
|
Antarctic Guide to Martian Weathering--- Soils in the Antarctic Dry Valleys contain traces of silicate alteration products and secondary salts much like those found in Martian meteorites.
Written by Linda M. V. Martel |
 |
Reference:
Insights from Polar Desert Soils
 |
The photograph on the left is a general scene of the environment of Wright Valley, Antarctica. The ice-free landscapes of the Dry Valleys seem out of place on this continent otherwise known for its permanent ice coverage. During the austral summer field season, temperatures in the Dry Valleys fluctuate around freezing. Mean temperatures are -23.7 oC to 0.7 oC. The photograph below is the specific soil pit in the Prospect Mesa of Wright Valley where soil samples were collected and studied by Wentworth and colleagues. For scale, the length of the metal tube on the pit wall is 78 centimeters.

|
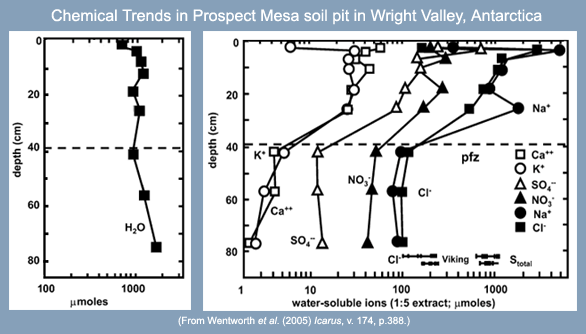 |
| Graphs showing the water content (left) and water-soluble ions (right) measured in soil samples from the Wright Valley soil pit. Below about 40 centimeters (indicated by the dashed line) the soil is permanently frozen. |
Wentworth and colleagues compare their results to those of several more recent Dry Valleys field studies by other research teams (e.g., Dickinson and Rosen, 2003). A characteristic common to Dry Valley soils in general is the presence of one or more salt-rich layers of typically the same kinds of salts: calcium carbonate, calcium sulfate, and magnesium sulfate. Soils from different locations in the Dry Valleys have different characteristics. Differences are found in the depths (beneath the surface) of the salt layers, in the number of salt layers in each soil, and also in the spatial distribution of the types of salts. It is clear that water and salt migration patterns are markedly different in different locations in the Dry Valleys, and one interpretation is that the differences are largely due to the amount of precipitation and the availability of surface or subsurface water at the sampling sites. Some soils, especially those at elevations higher than the Prospect Mesa soil pit, have occasional snow accumulations, while some locations in the valleys are affected significantly by summer meltwater. If only minute amounts water become available, however, only the most soluble salts will be dissolved and carried away; these salts would later be deposited elsewhere, resulting in the spatial segregation of different salt species. Where relatively abundant moisture is present, salts and water migrate downward from the surface and become concentrated at one specific horizon; in this case some recent researchers suggest that subsurface ice acts like a cold trap and results in the net gain of both salts and water within the soils.
Comparing Wright Valley Soils with Martian Meteorites
Wentworth and colleagues have teased out the compositional and textural details of the terrestrial soils using scanning electron microscopy (SEM) and transmission electron microscopy (TEM). They find that silicate grains have been altered by water throughout the Antarctic soil column, even in the permanently frozen zone. The images, below, show similarities between silicate alteration features in the Wright Valley soils and in the Martian meteorite Shergotty, which was collected in August of 1865 in India.
 |
| When silicate minerals dissolve they leave behind pits with sharp "sawtooth" margins or honeycomb patterns. Figures (a) and (c) show effects of aqueous dissolution on amphibole found in the permanently frozen soil collected in Wright Valley, Antarctica. Figures (b) and (d) show dissolution features on pyroxene in Shergotty. |
Trace amounts of salts, notably calcium carbonate, calcium sulfate, and magnesium sulfate, occur in the Wright Valley soils and also in Martian meteorites Chassigny [Data link from Meteoritical Database] and Nakhla [Data link from Meteoritical Database], as shown in the images below. Chassigny was collected in October of 1815 in France and Nakhla was collected in June of 1911 in Egypt.
 |
| Images (a) of calcite rhombs on quartz and (c) calcium sulfate, labeled "gyp" for gypsum, are both from the Wright Valley soil. Image (b) shows calcium carbonate grains (labeled carb) around a central calcium sulfate grain (labeled Ca-sulf) in Martian meteorite Chassigny. Image (d) of the Martian meteorite Nakhla shows calcium sulfate on pyroxene (labeled px). |
You might wonder if the alteration products and salts in these meteorites formed after the rocks landed on Earth and are a result of terrestrial weathering. Though it is true that some meteorites in the world's collections have been altered by terrestrial weathering, this is not the case with the Martian meteorites considered in this study by Wentworth and colleagues. First, Chassigny, Shergotty, and Nakhla were observed falls and were picked up soon after they landed, so there was little time for weathering in a terrestrial soil. Second, even if they had been weathered after landing, the meteorites were not exposed to an Antarctic-like environment, as they fell in France, India, and Egypt, respectively. Third and most important, the Martian origin of the alteration products and salts in Chassigny, Shergotty, and Nakhla have been proven unambiguously. For instance, the researchers see alteration features that have been heated or truncated by the fusion crust (the glassy coating remaining after the rock blazed through the Earth's atmosphere.)
The mineral assemblages in Martian meteorites, so similar in composition and abundance to those found in the Dry Valleys soils, tell a story about the interactions between fluids and rocks in the Martian crust. Planetary geologists and meteoriticists are reading these long and complicated weathering histories. It seems likely that many of the salts in Mars meteorites formed through evaporation of briny Martian water by processes similar to that proposed by John Bridges and Monica Grady (Natural History Museum, London), for example, for the salt minerals in Nakhla.

|
| Rocks in the "Columbia Hills" show evidence of past alteration by water as determined by the Mars Exploration Rover Spirit. [Click image for high resolution version and additional details.] |
Since aqueous processes are active even in the permanently frozen zones of Dry Valleys soils, Wentworth and colleagues postulate that similar processes are probably also occurring on Mars today, especially at the mid-latitudes or in the polar regions during their respective Martian summers. They say that if the analogy to Antarctica holds true, then local areas on the Martian surface will certainly have distinct weathering histories. In the Dry Valleys soils, weathering products and life are distributed heterogeneously. Hence, Wentworth and coauthors say that such variations should be taken into account in future studies of Martian soils and also in the search for possible life on Mars.
The valuable studies by Wentworth and colleagues on terrestrial analog soils and Martian meteorites are providing insights into how to best characterize the past and present weathering environments on Mars and how to search for life on the planet. Their work demonstrates the need for many samples of rock and soil from different climate regimes, from different locations within each regime, and from different depths beneath the surface of Mars.
|
|
[ About PSRD |
Archive |
Search |
Subscribe ] [ Glossary | General Resources | Comments | Top of page ] |