|
|
|
|
|
|
|
|

|
The Multifarious Martian Mantle
Written by G. Jeffrey Taylor |
References:
Samples of Martian Lava Flows
Much of the surface of Mars is made of volcanoes or lava plains, or sedimentary rocks made from them. River valleys and canyons dissect volcanoes and lava flows, and long ago, before about 4 billion years ago, chunks of asteroids and comets blasted the surface to make the cratered Martian highlands. Nevertheless, remnants of the lavas remain. More recent impacts flung bits of lava flows and other types of igneous rocks off the planet. Some of the ejected rocks made their way to Earth; we've found about 30 of them. These free samples of Mars are invaluable in helping us understand the geological history of the Red Planet.
The main evidence that the rocks actually come from Mars is from trapped gases in three of them. The mixture of gases is a striking match for the Martian atmosphere as measured by the Viking landers in the mid-1970s. All of the Martian meteorites share a unique oxygen isotopic composition, linking them to each other and to the three with trapped Mars air in them.
There are several groups of Martian meteorites. The most common, shergottites, are basalts, a common type of lava flow. The shergottites might be chips of thick surface flows or solidified magma that cooled in dikes below the surface. They have a wide range in chemical and mineralogical compositions. They tend to have much less plagioclase feldspar than do typical terrestrial basalts, but like basalts on all the planets they formed by partial melting of the interior. Quite a few processes can operate as a magma oozes through a hundred kilometers of rock to the surface, but we can often see through these processes-in fact, we can learn a lot about them-to deduce the chemical composition and much about the origin of the rocks making up the interior, or mantle. Lars Borg, Chris Herd, and their colleagues have been able to determine the time of initial differentiation of Mars, hence the time the mantle formed, and to deduce the variations in the composition within the mantle.
 |
Polished sample of a shergottite, Sayh al Uhaymir 005 [Data link from Meteoritical Database], which was found in Oman. The image is an x-ray map obtained in an electron microprobe. Light green is olivine, dark green is pyroxene (actually two different minerals, one higher in calcium than the other), and pink is maskelynite (plagioclase feldspar shocked impact to a glass). Meteoriticists call such olivine-rich shergottites "olivine-phyric shergottites" to distinguish them from shergottites (similar but with little or no olivine) and lherzolitic shergotittes (lots of olivine and pyroxene, little plagioclase, and probably not lava flows). |
Young Meteorites, Old Mantle
The shergottites come from lavas that erupted between about 150 million years and 500 million years ago. Nakhlites (pyroxene-rich rocks that formed in different types of lava flows from Shergottites) and the meteorite Chassigny are older, 1300 million years (1.3 billion years). The oldest Martian meteorite we have identified is Allan Hills 84001 [Data link from Meteoritical Database], which formed from a magma intruded deep in the Martian crust 4.5 billion years ago.
Although the shergottites are all quite young compared to the 4.55 billion years age of the planets, they nevertheless contain a clear record of the time when their mantle source regions formed. (Cosmochemists refer to the region in a planet's mantle that melted to give rise to a magma as the magma's source region.) Lars Borg and his colleagues show this in two ways. The first was to investigate the isotopes of rubidium (Rb) and strontium (Sr). This requires painstaking work in ultraclean laboratories to prevent contamination. The samples are crushed, minerals separated, and each separate dissolved in assorted acids, run through a process that separates Rb from Sr, and isotopes measured by mass spectrometry.
 LEFT: Lars Borg in the isotope clean lab at the Johnson Space Center.
LEFT: Lars Borg in the isotope clean lab at the Johnson Space Center.
Using their own and analyses from other cosmochemists, Borg and his colleagues plotted the 87Rb/86Sr ratio in each rock against the 87Sr/86Sr ratio (see graph below). 87Rb is radioactive and as time passes it decays to 87Sr. If all the mantle sources for the rocks had the same 87Sr/86Sr but different amounts of Rb and Sr, they would have formed a horizontal line initially. With time, 87Rb would decay to 87Sr, leading to a line with a slope. The slope corresponds to the age of the mantle sources. If the individual points all plot close to a single line, it suggests that they had a simple history: All their mantle sources (hence much, perhaps most of the Martian mantle) formed simultaneously at the time indicated by the slope of the line. Each source subsequently melted, but at different times, producing lavas 150 to 575 million years ago (the ages of the shergottites). The slope of the line drawn between the point for QUE 94201 [Data link from Meteoritical Database] and Shergotty [Data link from Meteoritical Database], which most of the other points lie close to, indicates an age of 4.49 billion years. (The points falling off the line are the nakhlites.) Note that the values of 87Rb/86Sr, which reflect the elemental ratio of Rb/Sr, range quite widely. I return to this important observation below.
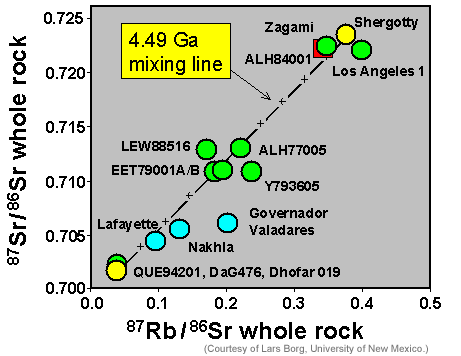 |
Whole rock Rb-Sr isotopic data for shergottites and nakhlites. The shergottites all fall along a line suggesting an ancient age of 4.49 billion years. This age may represent the time when distinct sources formed in the Martian mantle. |
Cosmochemists get age information from samarium (Sm) and neodymium (Nd), two rare earth elements. Sm has two radioactive elements. 147Sm decays to 143Nd with a long half-life of 106 billion years. On the other hand, 146Sm decays to 142Nd with a much shorter half-life, only 103 million years. 146Sm decays fast enough that it would be almost completely transformed to 142Nd in about five half lives, about 500 million years (0.5 billion years). The shergottites have clear evidence that this short-lived isotope was present in their mantle sources, so the sources formed before 4.0 billion years ago.
We have two speedometers here, one recording city driving, the other highway driving. Combined they ought to give us a more complete record of shergottite driving habits, i.e., the ages of their mantle sources. In December, 2002, Lars Borg was pondering these Sm-Nd isotopes systematics as he and his dog, Meka (who should have been named Isochron), were driving from New Mexico to his wife's parents' home in California. His wife and young child had flown to the west coast, leaving him alone with his thoughts. (Meka is not particularly talkative.) While driving through high, colorful deserts and low, hot, barren ones, Borg worked out that he could use both the long-lived and short-lived Sm isotopes to define the age of the shergottite mantle sources even better than the Rb-Sr data had, or at least make an independent assessment of it.
Borg realized he could use three parameters to assess the age of differentiation of shergottite mantle sources. One is the initial ratio of 143Nd/144Nd in each shergottite, expressed as epsilon-143Nd, a measure of how the ratio deviates from the ratio in chondritic meteorites, in parts per thousand. The other is the initial ratio of 142Nd/143Nd, or epsilon-142Nd, the deviation of the ratio from chondritic meteorites, again in parts per thousand. He also needed to know the ratio of 147Sm/144Nd in the source regions. Assuming that all the epsilon values were initially like those in chondrites, that formation of the sources involved formation of reservoirs with different Sm/Nd ratios, and that the sources melted only once more (when each shergottite magma formed), Borg knew he could calculate lines of equal age (isochrons) for different times. By plotting data from shergottites on the resulting complicated graph, Borg hoped to find the age of mantle differentiation.
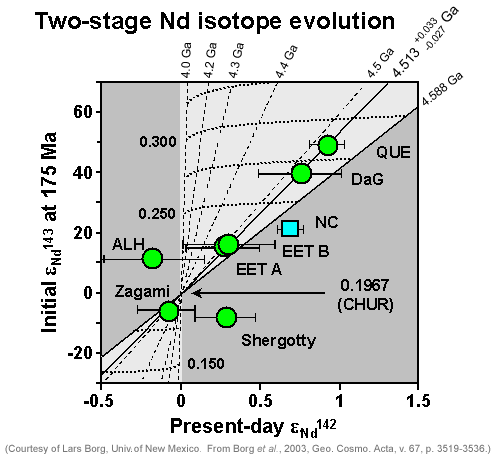 |
Lars Borg devised this plot of the variation in Nd isotopes with time and ratio of Sm/Nd after mantle formation, assuming a simple two-stage history for Martian meteorites (early mantle formation followed billions of years later by magma formation). The circles are for shergottites (except the one labeled "ALH," which is not a basalt) and the squares are for the nakhlites and chassigny. The filled circles fall along an isochron indicating an age of 4.51 billion years, which Borg and his colleagues take as the age of formation of the shergottite source regions. The points off the line, including Shergotty and the nakhlites, probably had histories more complicated than the simple two-stage history upon which the isochron lines are based. |
Distinctive Mantle Reservoirs
The isotopic data show that the mantle sources for the shergottites formed at about 4.5 billion years ago, soon after Mars formed. To retain a record of that important event, much of the mantle cannot have been disturbed until shergottite magma formation 150 to 500 million years ago. This suggests that, unlike the Earth, there was not extensive mixing of the mantle or recycling of the crust back into it, unless that took place before 4.5 billion years ago.
Other data allow Borg and his coworkers to identify other chemical properties of the mantle sources, or reservoirs. A distinctive characteristic of shergottites is that they have a range of rare earth element concentrations, especially in the concentration of light rare earth elements (those with lower atomic weights). This can be expressed in a shorthand way by the ratio of lanthanum (La) to ytterbium (Yb). Specifically, La/Yb correlates with initial 87Sr/86Sr (see diagram below) and with epsilon-143Nd. The samples with high La/Yb (about the same as in chondritic meteorites) come from "enriched" mantle reservoirs. Those with low La/Yb come from "depleted" reservoirs. The depleted reservoirs have low initial 87Sr/86Sr and high epsilon-143Nd. Scott McLennan (State University of New York, Stony Brook) also identified distinctive mantle reservoirs by comparing trace element concentrations in Martian meteorites.
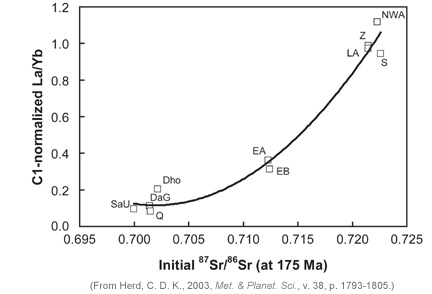 |
| Initial 87Sr/86Sr correlates with the extent to which La/Yb is depleted compared to chondritic (C1) values. Enriched sources have high 87Sr/86Sr because they had higher 87Rb/86Sr initially, hence produced more 87Sr by decay of radioactive 87Rb. |
Oxidation State of Mantle Reservoirs
The shergottites reveal another fundamental characteristic of the distinctive mantle reservoirs: the enriched reservoirs are significantly more oxidizing than the depleted ones. This is revealed by determination of a parameter called the oxygen fugacity, which is related to the partial pressure of oxygen available in a rock to oxidize elements that can occur in more than one valence state. Iron is particularly affected. Under highly oxidizing conditions it can be all Fe3+ (trivalent). Under somewhat reducing conditions it might be all Fe2+. When conditions are very reducing, much of the iron might be metallic (Fe0).
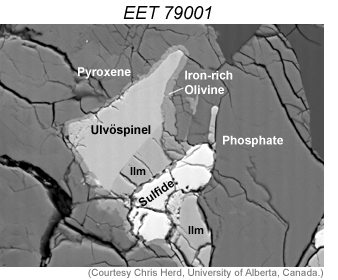 Cosmochemists determine oxygen fugacity by measuring the composition of iron-bearing minerals in a rock. Specifically, they determine the amount of Fe2+ and Fe3+ in each mineral. Examples are two iron-titanium oxide minerals (see photo on the left) or an iron-bearing oxide called spinel associated with pyroxene and olivine (also both iron-bearing minerals). It requires very careful observations of the mineralogy and the way minerals mingle in rocks to choose the right minerals to analyze. It also requires extremely accurate analyses by electron microprobe because the amount of Fe3+ cannot be determined directly-only total Fe can be measured. The amount of di- and tri-valent iron is determined by calculating the precise formula for a mineral and assuming the analysis is perfect (the total of all elements is exactly 100 percent).
Cosmochemists determine oxygen fugacity by measuring the composition of iron-bearing minerals in a rock. Specifically, they determine the amount of Fe2+ and Fe3+ in each mineral. Examples are two iron-titanium oxide minerals (see photo on the left) or an iron-bearing oxide called spinel associated with pyroxene and olivine (also both iron-bearing minerals). It requires very careful observations of the mineralogy and the way minerals mingle in rocks to choose the right minerals to analyze. It also requires extremely accurate analyses by electron microprobe because the amount of Fe3+ cannot be determined directly-only total Fe can be measured. The amount of di- and tri-valent iron is determined by calculating the precise formula for a mineral and assuming the analysis is perfect (the total of all elements is exactly 100 percent).
LEFT: The distribution of Fe3+ and Fe2+ between iron-titanium oxide minerals ilmenite and ulvöspinel can be used to determine oxygen fugacity. This shows co-existing oxide and other minerals in the shergottite EET 79001 [Data link from Meteoritical Database]. The image is a backscattered electron image taken with a scanning electron microscope. [Image is 150 microns across.]
It takes careful workers like Chris Herd, Cyrena Goodrich, and Larry Taylor (University of Tennessee) to do that type of work. Their hard work paid off. They found that oxygen fugacity also correlates with La/Yb (see diagram below). The enriched reservoir (high La/Yb) is more oxidizing than the depleted one, by a factor of about 1000. (The oxygen is usually reported in logarithm units of atmospheres, but often relative to the fugacity of a known equilibrium assemblage. In the plot below, from Chris Herd's paper, it is reported relative to the quartz-fayalite-magnetite (QFM) assemblage, or buffer.)
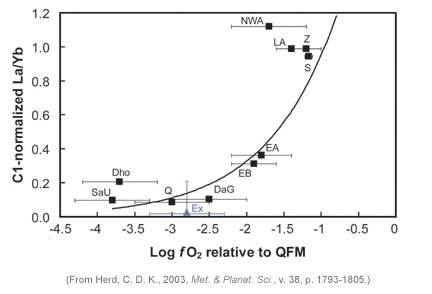
LEFT: Graph of La/Yb (normalized to the ratio in carbonaceous chondrites) versus oxygen fugacity (compared to the quartz-fayalite-magnetite QFM buffer assemblage). Enriched (high La/Yb) mantle sources are substantially more oxidizing than depleted (low La/Yb) mantle sources.
Formation of Mantle Reservoirs
Mars appears to have at least two distinct mantle reservoirs, and probably others. The table below summarizes the properties of the two distinct reservoirs tapped by magmas that created the shergottites.
| Enriched Reservoir |
high La/Yb low Sm/Nd (-εNd) high Rb (high 87Sr/86Sr) oxidized |
| Depleted Reservoir |
low La/Yb high Sm/Nd (+εNd) low Rb (low 87Sr/86Sr) reduced |
In papers published a couple of years ago, Borg, Herd, and company called the enriched reservoir "crust-like." This followed our thinking about planetary crusts being enriched in trace elements compared to the underlying mantles. It is also consistent with observations of the Mars Odyssey Gamma-Ray Spectrometer, which shows higher levels of K and Th (both elements that behave like rare earth elements) compared to Martian meteorites. However, placing the enriched component in the crust required that depleted shergottite magmas interacted with the crustal rocks to become more oxidized, enriched in trace elements and 87Sr/86Sr, and lower in Sm/Nd in very precise ways that are require disparate chemical properties and processes to precisely balance for each magma, an unlikely event. The solution was to put the reservoirs into the mantle.
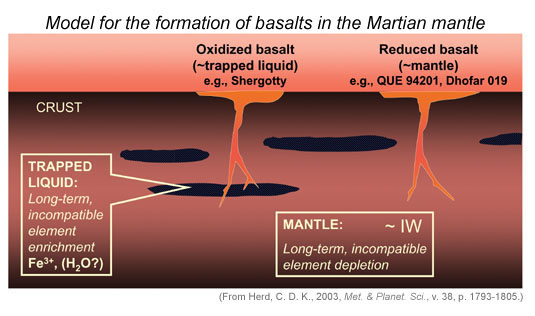 |
Model for shergottite (and Martian basalts in general) formation in the Martian mantle. Two reservoirs were produced early in Martian history. One is depleted in trace elements and is highly reducing (almost at the iron metal-iron oxide buffer "IW"). The other is enriched in trace elements and oxidizing. It contains substantially more Fe3+ than does the depleted reservoir, but might also contain H2O (though this is not necessary to cause the oxidation state to be higher than in the depleted reservoir). |
How did the reservoirs form? The very old age of the reservoirs drive Borg and others to suggest that Mars was surrounded by an ocean of magma when it formed, as most planetary scientists think happened to the Moon when it formed. [See magma ocean illustration in PSRD article The Oldest Moon Rocks.] If so, as the magma ocean began to crystallize, the first minerals would not take up much rare earth elements (including La, Sm, Nd, and Yb) or Rb. As crystallization continued the magma would become progressively enriched in elements excluded from the crystallizing solids, perhaps reaching values a hundred times higher. This could produce a large range from depleted mantle to highly enriched. And, because Fe3+ tends to be excluded from crystallizing minerals more than Fe2+ does, the ratio of Fe3+ to Fe2+ increases, making the enriched reservoirs more oxidizing. If H2O were present, it would also concentrate in the enriched reservoirs.
Lars Borg and Dave Draper, his colleague at the University of New Mexico, modeled this process quantitatively. Such calculations have been done to try to understand the lunar magma ocean. In the Martian case, the depth of the magma ocean matters a lot because the deeper the magma ocean, the higher the pressure near its base. The pressure difference results in different minerals crystallizing and those minerals differ in the extent to which they exclude or include trace elements. This research is still in its early stages, but the basic approach is leading to hypotheses testable by analyses of Martian meteorites.
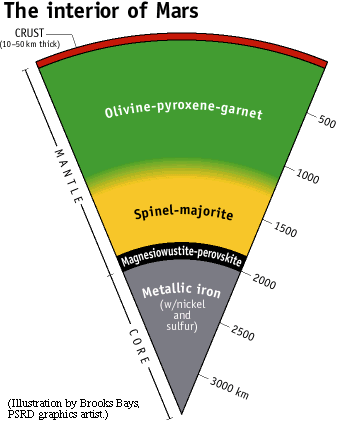 |
A likely picture of the interior of Mars, based on experiments by Constance Bertka and Yingwei Fei. The uppermost mantle of Mars consists of olivine and pyroxene, with a small amount of garnet (shaded green). These are fairly common minerals on Earth, the other planets, the Moon, and asteroids. However, at a depth of about 1100 km, the olivine begins to convert to a more dense form, called gamma-spinel, without changing its chemical composition. The conversion is complete by 1300 km. Along with the conversion of olivine to a spinel crystal structure, garnet and pyroxene convert to a mineral called majorite, which has a crystal structure like garnet, but is close to pyroxene in chemical composition (shaded yellow). At higher pressures, hence deeper, there is a relatively abrupt transition at 1850 km (shaded black) to a mixture of perovskite (itself a mixture chemically of MgSiO3 and FeSiO3) and magnesiowustite (a mixture of FeO and MgO). The metallic core (shaded gray) begins at about 2000 km depth and continues to the center at a depth of 3390 km. |
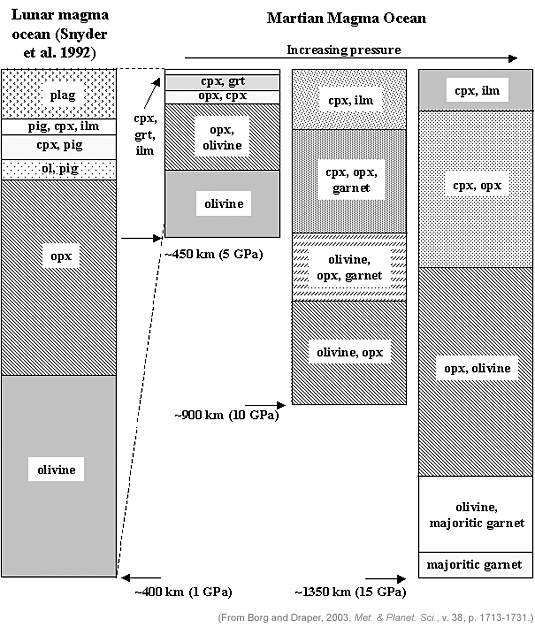 |
Cartoon showing the crystallization sequences for a lunar magma ocean (after Gregory Snyder, then at the University of Tennessee) and three Martian magma oceans with different depths. Higher pressure in the deeper oceans results in different element distributions. |
More Data Needed
It is astonishing that cosmochemists can determine so much about the formation of the Martian mantle from a few rocks that formed four billion years after the mantle did! Yet the unfolding story is reasonable. One test of the ideas will come from analysis of the composition of the Martian crust as determined by the Mars Odyssey Gamma Ray Spectrometer (papers are being written during June and July, 2004). The GRS data, particularly for the important trace elements K and Th, will allow us to compare to the compositions of shergottites and to assess how consistent the entire crustal composition is with the idea of enriched and depleted mantle reservoirs.
To fully assess the reservoirs, their ages, and models for their formation we need more samples, particularly basalts from the ancient highlands. We can hope to find more meteorites (the search for Antarctic meteorites now fields two teams each Antarctic summer), but we might still be stuck to whatever was blasted off Mars during the past few million years. Ideally, we will obtain more samples of Mars by sending robotic (and eventually piloted) spacecraft to Mars to return samples for study in labs here on Earth. Testing our ideas of mantle reservoirs and their formation requires the highly precise analyses that can be done only on earth, at least for now.
 |
NASA is tentatively planning to launch a sample-return mission to Mars in 2013. These paintings show two concepts for how the return vehicle will blast off. Note that both images have rovers. Recent experience with the Spirit and Opportunity rovers shows the need for mobility to collect the best possible samples. |
|
|
[ About PSRD |
Archive |
Search |
Subscribe ] [ Glossary | General Resources | Comments | Top of page ] |