|
|
|
|
|
|
|
 |
Moonbeams and Elements
Written by G. Jeffrey Taylor
Hawai'i Institute of Geophysics and Planetology
To determine how a planetary body formed and evolved, we must determine the chemical compositions of distinctive geologic regions on it. It is never possible to obtain enough samples of a planet to do this job thoroughly, so planetary scientists have searched for ways of determining chemical compositions from orbit, which would allow chemical mapping of the entire surface. A team at the University of Hawai'i has developed a method to determine the amount of titanium and iron on the lunar surface from the amount of sunlight reflected at different wavelengths. Most recently, David Blewett, Paul Lucey, B. Ray Hawke (University of Hawai'i), and Bradley Jolliff (Washington University in St. Louis) have used Apollo rock samples to carefully calibrate the technique, allowing surprisingly accurate measurements of iron and titanium. These two elements are especially useful in understanding the origin and geological evolution of the Moon.
You might be wondering, what can you learn from only two elements? It turns out that iron and titanium are quite informative. Titanium is an important probe of the compositions of magmas because it does not enter most minerals; instead it builds up in a magma during crystallization, so its abundance is a measure of the amount of crystallization that took place before a given rock formed in a large body of magma. Most important, the two elements allow us to distinguish among the known types of lunar rocks, and titanium forms the basis for classifying the basalts that make up the lunar maria.
 |
The main types of lunar rocks can be distinguished from one another by their iron and titanium concentrations. (In chemical analyses, these elements are expressed as oxides, FeO and TiO2, because they are chemically bonded to oxygen inside minerals.) Rocks from the highlands, the light-colored, rough, mountainous areas of the Moon, contain less FeO than do the maria, which are covered with dark lava flows. In fact, different groups within highland and mare rocks can be identified with just these two elements. |
There have been two recent missions to the Moon. The Galileo spacecraft flew by twice (in 1990 and 1992) as it was getting gravity assists for its trip to Jupiter (where it is now), and the Clementine spacecraft was in orbit for a few months in 1994. Both took pictures using special filters that allowed only certain wavelengths to pass through them. This produced images in several wavelengths, and it is the ratios of these wavelengths that allowed Blewett and Lucey to derive the elemental abundances. The Clementine mission obtained nearly complete global coverage at excellent spatial resolution; each pixel was about the size of a football field or two.
Deriving the FeO and TiO2 abundances involves plotting the intensity (called the reflectance) of light at one wavelength against the ratio of the reflectances at two wavelengths. This allows separation of the effects of "space weathering," which darkens the surface, from the effect of composition, which also darkens the surface. Without separation, it is difficult to distinguish one effect from the other. Space weathering is caused on the Moon by the combination of hydrogen being implanted by the solar wind (a stream of particles constantly emitted from the Sun) and impacts by tiny meteorites. The impacts melt some of the lunar regolith (the impact-produced, powdery layer on the Moon), while the hydrogen reacts with FeO to produce minute droplets of metallic iron.

|
These effects for the FeO technique are shown in the diagrams on the left. Continued space weathering causes an area of regolith to "mature" which makes it darker, as shown by the decrease in the reflectance at 750 nanometers (the x-axis). Space weathering also causes the regolith to increase in the ratio of reflectance at 950 nanometers to that at 750 nanometers,
which makes it spectrally redder. Different FeO concentrations in the regolith affect the properties in a different way than does space weathering. At a given FeO concentration, the space weathering moves the regolith along a line, as in the top diagram showing increasing maturity. However, increasing amounts of FeO in the regolith causes the whole array to pivot about a point, as shown in the bottom diagram. If you set up an x-y coordinate system with the pivot point at the origin, the angle q (theta) is related to the amount of FeO: higher q (theta) means higher FeO. |
A similar method using the reflectances at 415 and 750 nanometers was used to develop the technique for TiO2.
Samples of lunar regolith were returned by astronauts during the six Apollo missions and by automated spacecraft during three Russian Luna missions. Chemical analyses of those samples provide us with calibration points that we can use to improve the techniques for determining FeO and TiO2. The last three Apollo missions alone provide numerous calibration points as samples were collected at widely-separated localities because the astronauts were able to drive around in a dune buggy called the Lunar Roving Vehicle (LRV). In addition, those sites were geologically complicated, especially Apollo 15 and 17, where both mare and highland terrains were studied.
 |
This is a photograph of the Apollo 17 landing site in an area called the Taurus-Littrow Valley. The arrow indicates the location of the lunar lander. The circles show the sampling localities used by David Blewett and his colleagues to fine tune the technique used to determine the concentrations of FeO and TiO2. At each site, they averaged a group of nine Clementine pixels, making an area about the size of the circles. The sampling locations are in the centers of each circle. |
Blewett and his coworkers assembled a data base of chemical analyses of lunar regolith samples, and then used Clementine spectral data to determine the parameters, such as the angle in the diagrams above, used to calculate the FeO and TiO2 concentrations. They plotted these parameters against the actual concentrations, as determined from the returned samples. To optimize the data, they adjusted the pivot points on their spectral diagrams to improve the correlation of the parameter with actual FeO or TiO2 concentration. The resulting correlations are excellent, especially for FeO.
 |
 |
The scatter about the lines on these diagrams indicate an uncertainty in the FeO measurement of about 1 wt.% and an uncertainty in TiO2 of 1.1 wt.%. The uncertainties mean that measurements when either FeO or TiO2 are relatively low, say less than 2 wt.%, are not very accurate. For example, an apparent FeO concentration of 1 wt.% could correspond to an actual FeO content of between 0 and 2 wt.%. The scatter for TiO2 is more at concentrations less than a few percent. Lucey and Blewett believe that this might indicate that the variation of the titanium parameter with concentration may not be linear; instead, the correlation may be fit better by a curved line, rather than a straight one.
 |
This careful refinement could not have been done if we did not have lunar samples in our laboratories on Earth, and know precisely where on the Moon the samples were collected. |
Why did Blewett, Lucey, and their associates go to all this trouble? The answer is simple: the FeO and TiO2 contents of the lunar surface contain clues to how the Moon formed, the extent of its initial melting, the compositions of its lava flows, the mechanisms of crater formation, and other interesting facets of lunar history. They are applying their compositional tool to these problems, but most of those results are not yet published. When they are, the results will be reported in PSR Discoveries. For now, here are the outlines of some of their discoveries, based on the distribution of FeO on the Moon.
 |
The FeO concentration on the nearside (top image) and the farside (bottom image) of the Moon are shown in these images made from Clementine data and the FeO technique. The maria are obvious on the nearside image, showing up in red and yellow. On the farside image, there is a big green splotch, which marks the enormous South Pole-Aitken basin, an impact crater 2500 km across. It has higher iron than its surroundings, probably because it dug up the lower crust and mantle, which are probably higher in FeO than the highlands. (Image enlargement) |
Most of the farside and the highlands portions of the nearside are relatively low in FeO. Lucey and coworkers argued in the 1995 paper that this suggested fairly abundant aluminum in the Moon (in lunar rocks low iron correlates with high aluminum, that is, rocks with lower iron tend to have higher aluminum). In fact, they used their measurements to infer that the Moon contains more aluminum than does the Earth, ruling out the fission and binary-planet hypotheses for the Moon's origin. In fission, the primitive Earth spins fast, and a blob is flung off, forming the Moon. This theory implies that the rocky parts of the Earth and Moon have the same chemical compositions. In the binary planet hypothesis, the Moon forms in orbit around the growing Earth, as a two-object system from the start. Since both growing bodies would be fed by the same material, this idea also implies that the composition of the Earth and Moon are the same. Still in the running is the idea that the Moon formed as the result of the impact with the growing Earth of an object the size of Mars or larger, the so-called giant-impact hypothesis. In this idea, most of the Moon is formed from chunks of the huge projectile, which is likely to have a different composition than the Earth.
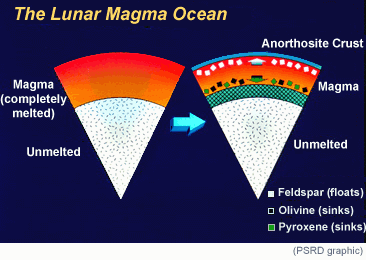 |
Rocks from the lunar highlands are relatively low in FeO and high in aluminum, which occurs mostly in the mineral plagioclase feldspar. In fact, some rocks, called anorthosites, contain almost nothing but plagioclase. This inspired some scientists to suggest that the highlands crust formed initially from a globe-encircling layer of molten rock, the lunar magma ocean, in which plagioclase floated and denser minerals sank. |
The idea of the magma ocean is a central theme in lunar science, and has been applied to other planets and some asteroids, but it is not a proven theory. How do we know that all the highlands are enriched in plagioclase (hence high in aluminum and low in FeO)? After all, we have samples from only a few places. Now, in a way, we have samples from the entire Moon, in the form of the Clementine data. The FeO image shows that most of the highlands are, as the magma ocean hypothesis predicts, low in FeO, and hence probably high in aluminum. So, it is likely that when the Moon formed it was surrounded by an ocean of magma, and the existence of that huge magma body may bear on the details of how the Moon was assembled after the giant impact that formed it.
All that, with more to come, figured out from some samples of lunar dirt and the light reflected from the Moon!
|
|
[ About PSRD |
Archive |
Search |
Subscribe ] [ Glossary | General Resources | Comments | Top of page ] |