Quick Views of Big Advances
Discovery of New Mineral, Krotite, in a CAI
 |
 |
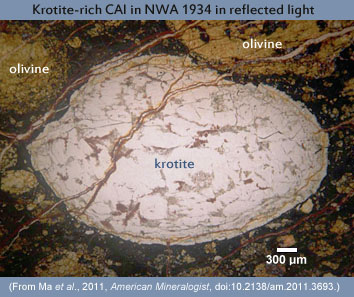
Krotite is the dominant mineral in the central and mantle areas of an unusual CAI in the NWA 1934 carbonaceous chondrite [Data link from the Meteoritical Bulletin]. The 2.75 mm x 4.5 mm inclusion is composed mainly of aggregates of (colorless and transparent) krotite crystals, with a few other calcium-aluminum or magnesium-aluminum oxides and a few silicates in a thin rim of concentric layers. Cracks, mainly filled with iron and aluminum hydroxides, crosscut the CAI's rim and parts of its interior giving it a "cracked egg" appearance. The CAI itself is surrounded by a matrix of mostly fine-grained olivine.
The discovery team suggests formation in a highly refractory condensate/evaporative environment in the cooling nebular gas. They say that the primary mineral assemblage in this CAI was introduced into a hot gas, which did not melt the CAI but perhaps reacted with surficial krotite crystals to produce the observed layered rim. The cracks may have been caused by compaction during accretion of the parent body, but the meteorite as a whole is essentially unshocked. A final, probably terrestrial, alteration process most likely filled the cracks with hydrated oxides during exposure to the northwest African environment. This CAI, along with its new mineral, krotite, will be the subject of additional research to determine formation and cosmochemical details. Because CAIs were the first solids formed in the solar nebula about 4.6 billion years ago, they help cosmochemists piece together records of nebular and early Solar System processes and how the first solid building blocks eventually turned into asteroids and planets.
![]() (pdf version)
(pdf version)
See:
Ma, C., Kampf, A. R., Connolly, Jr., H. C., Beckett, J. R., Rossman, G. R., Sweeney Smith, S. A., and Schrader, D. L. (2011) Krotite, CaAl2O4, A New Refractory Mineral from the NWA 1934 Meteorite, American Mineralogist, v. 96, p. 709-715. doi:10.2138/am.2011.3693.
Ma, C., Kampf, A. R., Connolly, Jr., H. C., Beckett, J. R., Rossman, G. R., Sweeney Smith, S. A. and Schrader, D. L. (2010) Krotite, IMA 2010-038. CNMNC Newsletter, October 2010, page 901; Mineralogical Magazine, v. 74, p. 899-902. [ pdf link ]
and the
PSRD article
Dating the Earliest Solids in our Solar System.
Written by Linda Martel, Hawai‘i Institute of Geophysics and Planetology, for PSRD.
|
|
[ About PSRD | Archive | CosmoSparks | Search | Subscribe ] [ Glossary | General Resources | Comments | Top of page ] |
