|
|
|
|
|
|
|
 |
A striking feature in the compositions of the Earth and Moon is their identical abundances of oxygen isotopes. Most planetary scientists agree that the Moon formed as the result of a giant impact with the proto-Earth. It explains some important characteristics about the Earth and Moon, such as why the Moon has a small metallic iron core, but planetary formation models suggest that the Moon ought to have a different oxygen isotopic composition than the Earth. Why is it the same? Kaveh Pahlevan and David Stevenson (Caltech) suggest that after the giant impact and before the Moon formed, Earth exchanged materials with the disk of magma and gas surrounding it, ironing out differences in their isotopic compositions. The process would take a couple of hundred years, about the time (100 to 1000 years) the disk would last before coalescing into the Moon. This interesting idea is far from proven, but it will undoubtedly lead to additional work because it has such great implications for the compositions of the Earth and Moon, when Earth received its water, and how planets accreted from the cloud of gas and dust surrounding the primitive Sun.
Reference:
The Cosmochemical Measure of the Moon
Cosmochemists do not agree completely about the differences and similarities between the bulk compositions of the Earth and Moon. They are sure that the Moon has a much smaller metallic core than does the Earth (see PSRD article: The Moon at its Core), so any model for the Moon's origin has to account for that important fact. The Moon is also depleted compared to Earth in volatile substances, those materials that boil at relatively low temperature. This includes water, but also moderately volatile elements such as potassium. This is shown by the ratio of potassium to thorium in the Earth (2,800) and Moon (360). Everybody agrees about the volatile depletion, too.
A particularly important point of agreement is that the silicate (rocky) Earth and Moon have identical (within analytical uncertainties) oxygen isotope compositions. There are three isotopes of oxygen, 16O, 17O, and 18O; 16O is most abundant. Plotting the ratio of 17O to 16O versus 18O to 16O shows that the data for both Earth and Moon fall on the same line. The line slopes in the way expected for chemical processing (crystallization, melting, alteration by water, and other processes). Data for meteorites, however, fall on other lines, indicating differences in the abundance of 16O. This is taken to be evidence that oxygen isotopes were not distributed uniformly throughout the solar system. Martian meteorites are distinctly different from lunar and terrestrial samples, containing less 16O than the Earth-Moon system. Other igneous meteorites contain more 16O than does the Earth-Moon system.
These relationships are shown in the diagram below. This diagram plots Δ 17O versus δ 18O, a diagram that isotope experts are particularly fond of. The δ 18O is simply the ratio of 18O to 16O, but normalized to the Earth value as represented by standard mean ocean water (SMOW). The Δ 17O (called "big delta O-17" by cosmochemists) is simply a measure of the vertical displacement of any point from the terrestrial fractionation line on a plot of δ 17O versus δ 18O. Anything exactly like the Earth falls on the dashed line. Note that all the points for the Moon fall on that line, indicating that the Moon has the same oxygen isotopic composition as does the Earth. Mars is way different, having much higher big delta O-17. This suggests that the relative abundances of the oxygen isotopes vary with distance from the Sun, or at least vary throughout the inner solar system.
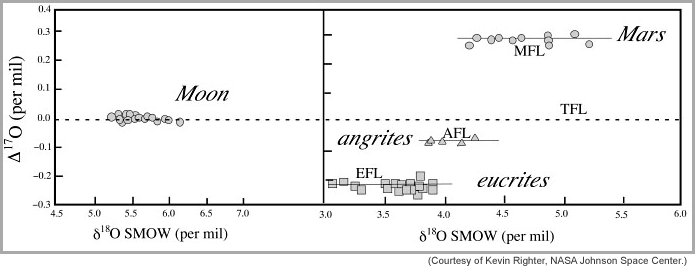 |
Precise oxygen isotope data for Earth (dashed line), Moon, Mars, asteroid 4 Vesta (eucrite meteorites), and the igneous angrite meteorites. The data for each body spreads out along the x-axis because of chemical processing, but all lie close to a line with distinctive big delta O-17. This shows that oxygen isotopic compositions vary throughout the solar system. TFL, MFL, AFL, and EFL stand for terrestrial, Mars, angrite, and eucrites fractionation lines, which is simply the way the isotopes vary with melting and crystallization. |
The similarity between terrestrial and lunar oxygen isotopes, in light of the variation throughout the solar system, has led many cosmochemists to conclude that the Earth and Moon must have formed from the same reservoir of material in the solar nebula. As explained below, however, our current understanding of how the inner planets accreted suggests that this is unlikely. So why do the Earth and Moon have the same oxygen isotopic composition? Pahlevan and Stevenson might have an explanation.
Cosmochemists do not agree about everything concerning the compositions of the Earth and Moon. Two particularly controversial issues are the abundances of refractory elements - elements that boil at high temperatures and hence condense first from a gas of solar composition, such as thorium, uranium, rare earth elements, and zirconium. Some of us think the Moon is enriched compared to the Earth in refractories, while others think the inventory of refractories is the same in the Moon and Earth (see PSRD article: Two Views of the Moon's Composition). Another bone of contention is the amount of oxidized iron (FeO) in the Moon compared to Earth. Some of us think the Moon is enriched (about 13 wt% FeO compared to only 8 wt% in the Earth), while others think the two bodies have the same concentrations of FeO.
Planetary dynamicists tell us the following story about planet formation: When the Sun formed it was surrounded by a disk of dust and gas. Planet formation began when dust grains clumped together to form, eventually, objects up to a few hundred kilometers across. Those hefty rocks rapidly accreted (taking maybe only a hundred thousand years) to form planetary embryos, which were roughly Moon to Mars-sized planetesimals. The planetary embryos took a more leisurely tens of millions of years to whack into each other and gather up left over smaller objects to form the inner planets.
 |
During the final stage of assembly of the inner planets, large planetary embryos accreted to each other, as depicted in this painting by James Garry. All were of substantial size (Moon to Mars size), so the accretionary impacts must have caused a lot of heating. The Moon formed from one such event in which the impactor was offset just the right amount to hurl melted and vaporized rock into orbit. |
The step from small planetesimals to planetary embryos was fairly orderly, with each embryo forming in its own relatively narrow feeding zone. Their compositions, therefore, reflected the composition of the dust in that region of the solar disk, and two embryos growing side-by-side would have had similar compositions. The accretion of planetary embryos was much more disorderly. Planetary embryos interacted with each other and with the large outer planets (if they had formed), causing changes to their orbits. The result was that when they collided, they did not necessarily come from the same neighborhood of the solar disk. This process has been modeled quantitatively by many investigators, including John Chambers (Department of Terrestrial Magnetism, Carnegie Institution of Washington), who provided the simulation shown below. Note how the growing planets become smaller in number and bigger in size. Most important, the embryos migrate inwards and outwards before accreting. This causes considerable shuffling throughout the inner solar system and even two planets that grow in the same region can have different compositions.
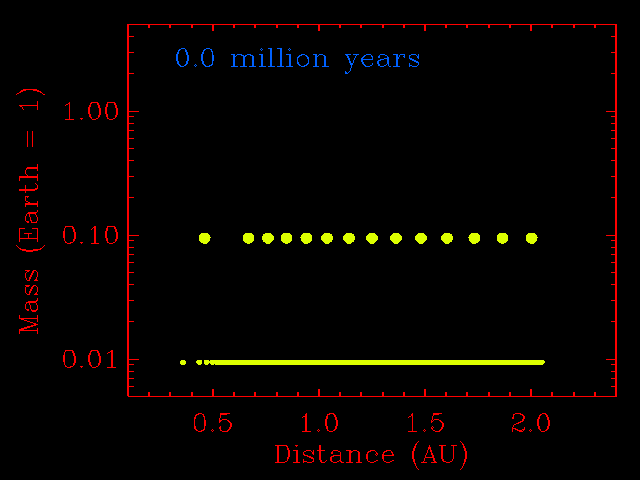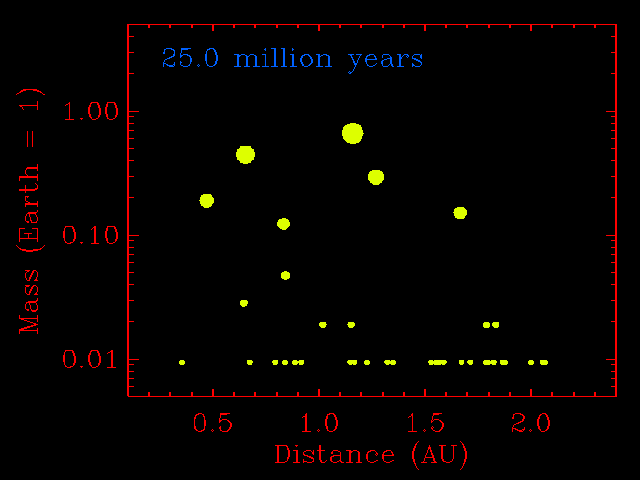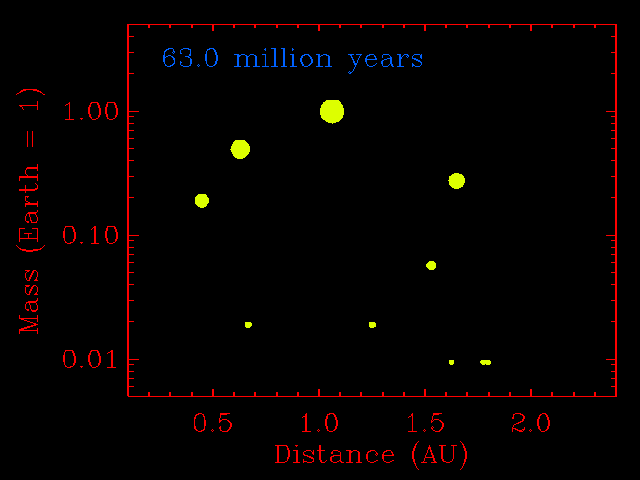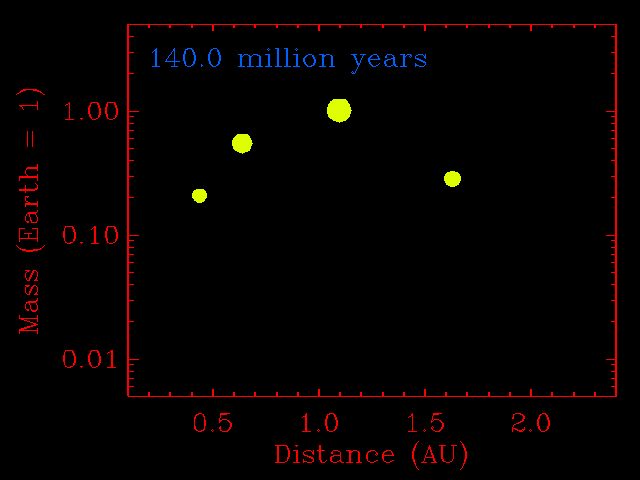 |
This physics-based simulation of the accretion of the inner planets from planetary embryos begins with a set of 14 embryos the size of Mars, and numerous planetesimals the size of big asteroids (1% of the Earth's mass). The x-axis is distance from the Sun in AU. The simulation assumes that objects that hit each other accrete, rather than bounce away. As the simulation proceeds (time marker at top left) the embryos do not assemble into a planet in a narrow zone. They hop around with abandon. This causes shuffling throughout the inner solar system and even two planets that grow in the same region can have different compositions. Simulation courtesy of John Chambers. |
 |
Summary of four of John Chambers' numerical simulations. They indicate that there was mixing of planetary embryos. The pie diagrams show the percentage of material in each of the final inner planets that came from the region of the solar nebula shown by the colors. Although most of the material for a given planet comes from relatively nearby, quite a bit comes from much farther away. If there was a gradient in the oxygen isotopic composition throughout the inner solar system, it is difficult to see how the Earth and Moon could end up with the same proportions of the three oxygen isotopes. |
All this mixing is a big deal when considering why the oxygen isotopic composition of the Moon is the same as the Earth's. Planetary scientists generally think that the Moon formed by a giant impact of a Mars-sized planetary embryo into the proto-Earth. Most of the Moon was constructed from melted rock and vapor ejected from the impactor, rather than the proto-Earth. The similarity in oxygen isotopic composition between Earth and Moon would seem to suggest that the impactor and proto-Earth had the same oxygen isotopic composition. The problem is that, if the simulations are correct, accretion processes mix stuff from all over the inner solar system, and even an impactor that formed in Earth's vicinity can have a different composition.
Kaveh Pahlevan quantified this by calculating from one of John Chambers' simulations the compositional scatter of accreting planetary embryos. Pahlevan assumed that the oxygen isotopic composition varies linearly with distance from the Sun, calibrating the line with the value of Earth and Mars (represented by Martian meteorites). Pahlevan and Stevenson found that none of the impactors had a composition as similar to the target proto-planet as we see for the Earth and Moon. The calculation, as Pahlevan and Stevenson point out, is not perfect as we do not know how oxygen varies throughout the inner solar system. On the other hand, we do know it varies. Assuming that the moon-forming impactor and the proto-Earth were different in oxygen isotopic composition, how did Moon and Earth end up with identical oxygen? Perhaps, Pahlevan and Stevenson suggest, the compositional variations were evened out in the hot environment accompanying the giant impact that formed the Moon.
The Scorching Aftermath of the Giant Impact
Giant impacts between planetary embryos are incredibly energetic. The proto-Earth and perhaps the Mars-sized impactor had already grown by large impacts, so were probably already hot. The Moon-forming event would have substantially melted the Earth and the ejected material would have consisted of molten rock, forming an orbiting magma disk, surrounded by an atmosphere of vaporized rock. Gravitational forces would have sheared off the connection between the orbiting magma and the molten Earth, but the hot (over 2000 Kelvin) silicate atmosphere would have been continuous. The idea is shown schematically in the diagram below.
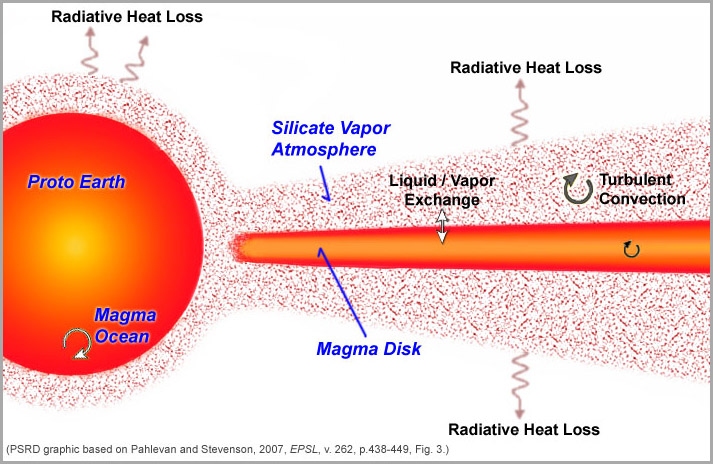 |
The proto-Earth and the proto-lunar disk immediately after the giant impact, as depicted by Pahlevan and Stevenson, based on a concept Dave Stevenson published in 1987. Rapid loss of heat into space causes convection in the Earth, the magma disk, and the silicate atmosphere, all of which facilitate exchange of material to even out differences in isotopic and chemical compositions. The silicate atmosphere would act as the exchange medium between Earth and the proto-lunar disk. Kaveh Pahlevan quantified the exchange processes. |
The hot proto-lunar disk surrounding the equally-hot Earth loses heat at the top of the silicate atmosphere. Pahlevan and Stevenson calculate from heat equations that this might take only 100 to 1000 years, with 1000 years being an extreme upper limit. This is all the time available to equal out the oxygen isotopic compositions of the disk (mostly from the impactor) and the proto-Earth. To see if that is feasible, Pahlevan and Stevenson calculated rates of convection, magma-vapor exchange, Earth-to-disk exchange, and radial mixing within the disk. In all but the last case, the rate of exchange is rapid, only weeks in most cases.
The rate at which the disk mixes radially is the slowest by far. This is important because the Moon forms from the outer part of the disk, which has a radius equal to about five times the Earth's radius. Using fluid dynamic principles, Pahlevan and Stevenson show that turbulent convection in the atmosphere could tap the entire terrestrial magma ocean, thereby equilibrating the isotopic compositions, in about 100 to 250 years. This is enough time to make identical oxygen isotopic compositions if the disk lasts 100 to 1000 years.
There are uncertainties in the calculations, and Pahlevan and Stevenson are not shy about listing them. One is that the outer part of the disk where the Moon forms might have cooled faster, hence not participated as fully in the equilibration process. Another is that rotation of the disk might inhibit radial diffusion in the disk (the important rate-limiting process). Nevertheless, the calculations certainly suggest that mixing between Earth and proto-lunar disk is possible.
Pahlevan and Stevenson's work assumes that our current ideas of planet formation are correct--that there was considerable mixing throughout the inner solar system and that the Earth and Moon began with different compositions, hence the need for an equilibration mechanism like the one they explore. However, as they point out, it is also possible that planet-formation models are flawed. For example, there are collisions that do not accrete, which violates an assumption of the simulations; see PSRD article: Hit and Run as Planets Formed for an example of one type of non-accretional event. Planet-formation models also do not take into account the effect the countless small bodies still lingering around the inner solar system might have on the orbits of planetary embryos, or if the small amount of gas left in the nebular disk has an effect on embryo accretion.
Pahlevan and Stevenson point out a few geochemical consequences of their mechanism. The equilibration implies that the Earth and Moon should have the same chemical composition as well as isotopic composition, but the authors note that other processes, such as condensation of the vapor to magma, might have caused separation of elements from one another. There are a variety of chemical effects in this complicated environment that need to be evaluated in detail.
We also need to really figure out what the Moon is made of. There are great uncertainties in our knowledge of the chemical composition of the Moon (see PSRD article: Two Views of the Moon's Composition). It might be enriched in refractory elements such as uranium and the rare earths compared to Earth, or not be. It might have a higher concentration of iron oxide in its rocky portions than does the Earth, or it might have the same as the Earth. Cosmochemists and other planetary scientists have their work cut out for them!
Fortunately, help is on its way. There are five orbital missions to the Moon. Two are in orbit now (the Japan Aerospace Exploration Agency's KAGUYA (SELENE) mission and the Chinese National Space Administration's Chang'e mission), and two will be launched in 2008 (the Indian Space Research Organization's Chandrayaan-1 mission and the NASA Lunar Reconnaissance Orbiter). All will measure the composition and other properties of the surface in considerable detail. The fifth, Grail, has just been selected by NASA. Grail will use highly accurate measurements of gravity to determine the interior structure of the Moon, from crust to core.
|
|
[ About PSRD |
Archive |
Search |
Subscribe ] [ Glossary | General Resources | Comments | Top of page ] |